Which Subshell Is Represented by the Lanthanides Family
Ion-exchange separations can also be made past the utilise of a polymer with exchangeable anions in this instance, the lanthanide or actinide elements must be initially nowadays as complex ions (11,12). The anion-exchange resins Dowex-one (a copolymer of styrene and divinylben2ene with quaternary ammonium groups) and Amherlite IRA-400 (a quaternary ammonium polystyrene) have been used successfully. The social club of elution is frequently the opposite of that from cationic-exchange resins. [Pg.215]
| Table 3. Electronic Configurations for Gaseous Atoms of Lanthanide and Actinide Elements... |  |
| Tabular array 9. Ionic Radii of Actinide and Lanthanide Elements... |  |
There is no single best form of the periodic table since the choice depends on the purpose for which the table is used. Some forms emphasize chemical relations and valence, whereas others stress the electronic configuration of the elements or the dependence of the periods on the shells and subshells of the diminutive structure. The most convenient form for our purpose is the so-called long form with separate panels for the lanthanide and actinide elements (meet inside front cover). There has been a lively debate during the past decade as to the best numbering system to be used for the individual... [Pg.xx]
The redox behaviour of Th, Pa and U is of the kind expected for d-transition elements which is why, prior to the 1940s, these elements were commonly placed respectively in groups 4, five and half-dozen of the periodic tabular array. Behaviour obviously like that of the lanthanides is not evident until the second one-half of the series. Withal, even the early on actinides resemble the lanthanides in showing close similarities with each other and gradual variations in properties, providing comparisons are restricted to those properties which practise not entail a change in oxidation state. The smooth variation with atomic number found for stability constants, for instance, is like that of the lanthanides rather than the d-transition elements, equally is the smooth variation in ionic radii noted in Fig. 31.four. This last factor is responsible for the shut similarity in the structures of many actinide and lanthanide compounds especially noticeable in the 4-3 oxidation state for which... [Pg.1266]
1 proper noun, more than than whatsoever other, is associated with the actinide elements Glenn Seaborg (1912-1999). Between 1940 and 1957. Seaborg and his team at the University of California, Berkeley, prepared nine of these elements (at no. 94-102) for the get-go fourth dimension. Moreover, in 1945 Seaborg made the revolutionary suggestion that the actinides, like the lanthanides, were filling an f sublevel. For these accomplishments, he received the 1951 Nobel Prize in chemistry. [Pg.147]
Unsubstituted bisphthalocyanines 2 are formed in the presence of several elements which exist in a stable oxidation state of + III or +IV such as titanium, zirconium, hafnium, indium and most of the lanthanide and actinide elements. [Pg.751]
Unusual oxidation states of some actinide and lanthanide elements. 50. B. Asprey and B. B. Cunningham, Prog. Inorg. Chem., 1960, 2, 267-302 (245). [Pg.26]
| Figure 3. The lattice parameter for the family of stone-common salt structure actinide-antimonide compounds is shown where the line is for the corresponding lanthanide compounds. The metal radii for the calorie-free actinide elements are plotted. The smooth line simply connects Ac to the heavy actinides. In both cases the smooth line represents the ideal tri-valent behavior. | 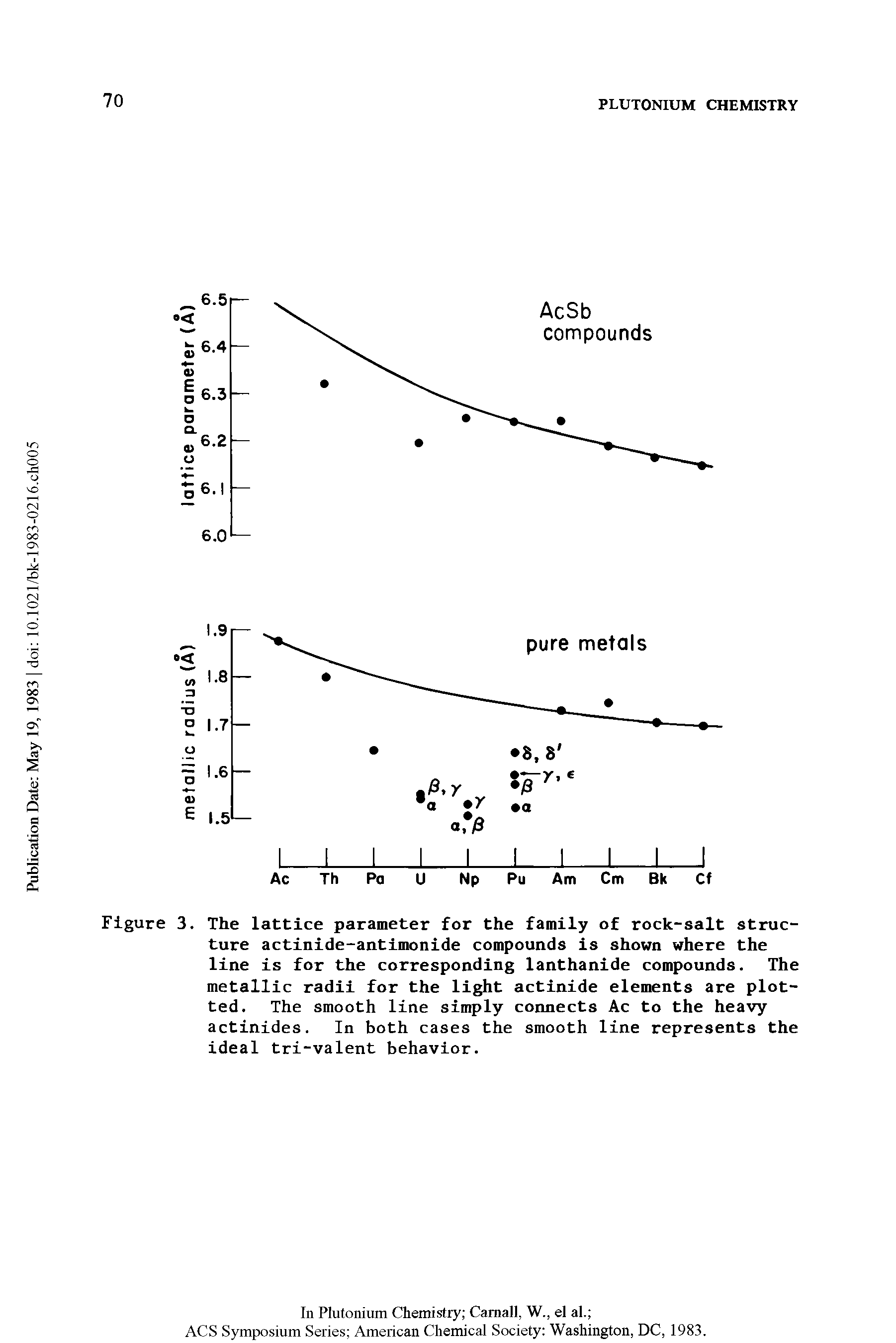 |
Webb LM, Taylor DM, Williams DR. 1998. Computer modeling of the chemical speciation of lanthanide and actinide elements in the man gastrointestinal tract Mouth and tum. Radiat Prot Dosim 79(50/4) 219-222. [Pg.266]
Equally the atomic number increases, then does the positive charge of the nucleus, and the electrons are bound with a higher energy. However, this increase is non linear. For case, the electrons in the d orbital of the third beat out take a higher energy than those in the s orbital of the quaternary shell, and hence the latter are filled outset. The effect is the unexpected behavior of the first ten transition elements. In the case of the actinides and lanthanides, even more inner orbitals are occupied. Nature is not so simple, only the scheme should help to visualize this circuitous structure. And if one can assign the electrons of an chemical element, one is a step closer to successfully unraveling the mysteries of the Periodic Tabular array. [Pg.112]
As to the first route, we started in 1969 (i) in investigating unconventional transition metal complexes of the 5 and 4f block elements of periodic table, e.g., actinides and lanthanides as catalysts for the polymerization of dienes (butadiene and isoprene) with an extremely high cis content. Even a small-scale increment of cistacticity in the vicinity of 100% has an important effect on crystallization and consequently on elastomer processability and backdrop (two). The f-block elements have unique electronic and stereochemical characteristics and give the possibility of a participation of the f-electrons in the metal ligand bond. [Pg.34]
Lanthanide elements are /-block elements in which the four /-subshell is filling Ce through Lu. Actinide elements are in which the 5 /-subshell is filling Th through Lr. [Pg.184]
Asprey, Fifty. B. and Cunningham, B. B., Unusual Oxidation States of Some Actinide and Lanthanide Elements. 2 267... [Pg.626]
Figure five.13. Scheme of correspondence between the different lanthanide and actinide elements which may be causeless on the basis of several similarities and analogies in their chemical and alloying behaviours. [Pg.382]
Wish, L., E. C. Freiling and R. Bunney Ion-Exchange equally a Separation Method. 8. Relative Elution Position of Lanthanide and Actinide Elements with Lactic Acid Eluant at 87° C. J. Amer. chem. Soc. 76, 3444 (1954). [Pg.20]
Lawrencium is the terminal of the transuranic elements and the xv th in the actinide series (in that location are fifteen elements in the lanthanide serial likewise, assuming you lot starting time counting the series at the elements lanthanide and actinium, respectively.) It is assumed that lawrencium has some chemical and physical characteristics similar to lutetium, located just above it in the lanthanide series. It is also located at the bottom of the group 17 (VILA) elements, which makes it the heaviest of the halides. [Pg.336]
The actinide elements result from the Ailing up of the bf orbitals and are the analogs of the lanthanides (J). There is a shut similarity between the trivalent 4/and 5/ions. However, the actinides differ from the lanthanides in... [Pg.381]
The review is divided into three sections, (i) the chemical science of scandium and yttrium, (ii) the chemistry of the lanthanide elements, farther subdivided into a survey of lanthanide due north.m.r. shift reagents and the full general chemistry of the elements, and (iii) the chemical science of the actinide elements and uranyl and related compounds. [Pg.451]
Americium may be separated from other elements, particularly from the lanthanides or other actinide elements, by techniques involving oxidation, ion substitution and solvent extraction. One oxidation method involves precipitation of the metal in its trivalent state every bit oxalate (controlled precipitation). Alternatively, information technology may be separated past precipitating out lanthanide elements as fluorosilicates leaving americium in the solution. Americium may also he oxidized from trivalent to pentavalent state past hypochlorite in potassium carbonate solution. The product potassium americium (V) carbonate precipitates out. Curium and rare earth metals remain in the solution. An alternative approach is to oxidize Am3+ to Am022+ in dilute acid using peroxydisulfate. Am02 is soluble in fluoride solution, while trivalent curium and lanthanides are insoluble. [Pg.17]
Americium and other actinide elements may be separated from lanthanides by solvent extraction. Lithium chloride solution and an viii to ix carbon third amine are used in the process. Americium is and then separated from curium by the above methods. [Pg.18]
Molecular Interactions Reaction Kinetics Bones Spectroscopy X-ray Crystallography Lanthanide and Actinide Elements Maths for Chemists Bioinorganic Chemistry Chemistry of Solid Surfaces Biology for Chemists Multi-element NMR... [Pg.150]
Source: https://chempedia.info/info/actinide_elements_lanthanides/
0 Response to "Which Subshell Is Represented by the Lanthanides Family"
Post a Comment